
Natural killer cells (NK cells) are a group of immune cells with a CD3-CD56+ phenotype, which are usually found in lymph nodes, various organs and peripheral blood. NK cells account for about 5-20% of the peripheral blood lymphocytes of the human body, thus the peripheral blood is one of the suitable sources for obtaining NK cells. Due to the insufficient number and cytotoxicity of NK cells collected directly from the peripheral blood, therefore it is necessary to increase the number and cytotoxicity of NK cells by ex vivo expansion before being used for treatment.
An 11-year follow-up epidemiologic survey involving more than 3,600 adults has shown that the extent of NK cell activity in peripheral blood is negatively associated with cancer risk, and this phenomenon is true for both men and women. The presence of tumor-infiltrating NK cells has been regarded as one of the positive prognostic factors for a variety of malignant tumors, including colorectal cancer, gastric cancer and squamous cell lung cancer (5-7). Studies have shown that NK cells have the potential to treat solid tumors; and the results of many recent studies have shown that NK cells can also be used to treat hematological malignancies, including Acute Myeloid Leukemia (AML) (8-9) and Multiple Myeloma (10-11) and so on.
NK cells can kill cancer cells (1-3) through the following mechanisms: (i) secreting perforin and granzyme to induce apoptosis of cancer cells. (ii) the death ligand binds to the death receptor on the surface of cancer cells to trigger the apoptosis of cancer cells. (iii) secreting cytokines such as TNF-α and IFN-γ to regulate the functions of immune cells, such as T cells and dendritic cells, thereby strengthening the anti-cancer ability. (iv) in the presence of antibody, the FCγRIII (CD16) of NK cells binds to the Fc portion of antibody triggering the lysis of targeted cells. This phenomenon is also known as antibody-dependent cell-mediated cytotoxicity (ADCC).
Medigen has developed a specialized methodology to expand and activate NK cells from small amount of peripheral blood. The expanded NK cells produts based on our technology are called the “Magicell-NK”, which have large numbers of cells, high purity, high cytotoxicity, and does not use any non-human source ingredients and feeder cells.
After 14 ± 2 days of in vitro expansion, Magicell-NK consists of cells having a phenotype of CD3-CD56+ NK cells for more than 95%, which are considered as the high-purity NK cells. The overall cell survival rate is greater than 98%.
Furthermore, the killing ability of Magicell-NK on cancer cells can reached up to 90% (Figure 1).
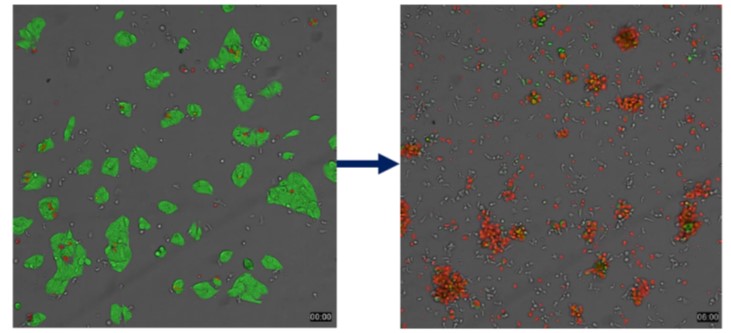
Figure 1. Magicell-NK has high cytotoxicity.
(Note: The green color on the left showed the liver cancer cells (HepG2) that were alive before killing; the red color on the right showed liver cancer cells that have died which were killed by Magicell-NK. The smaller colorless dots are Magicell-NK.)
References: